Qualityze
- 19 profiles
- 18 categories
Featured Products
All Products & Services
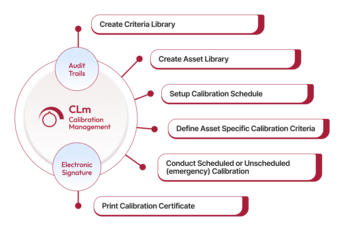
Qualityze Calibration Management allows organizations to manage their critical assets by streamlining calibration programs while complying with regulatory requirements. It seamlessly integrates with your existing quality processes.
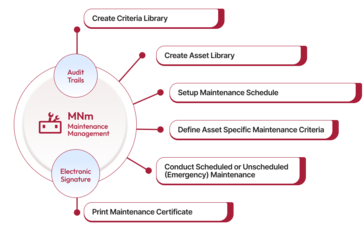
Every organization needs a standardized maintenance management system that can help manage production instabilities, malfunctioning machines, or complete breakdowns to avoid potential pauses in the production, wholly or partially. Qualityze Maintenance Management Solution allows organizations to create and manage the maintenance programs to ensure the safe functioning of the equipment and hassle-free production. You can integrate Qualityze with an existing quality management system to streamline the maintenance activities more efficiently.
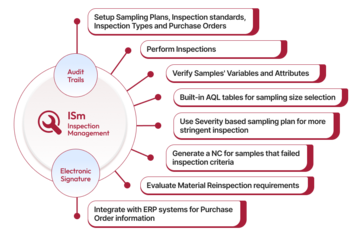
Qualityze Inspection Management streamlines the end-to-end process for incoming, pre-shipment, and production inspections, managing compliance risks effectively. It fosters a culture of continuous improvement by ensuring prompt reviews. Its advanced reporting tools offer quality teams improved visibility into inspections, ensuring that incoming materials or components adhere to the latest standards and guidelines.
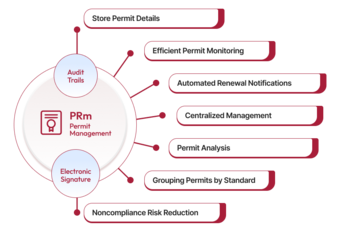
The permit management software facilitates the process and helps maintain your positive reputation, goodwill, and good public relations in today’s environment of compliance and transparency. It also prevents delays related to paperwork, which can arise when opening new facilities or launching new products, as different types of businesses and activities are subject to various licensing, permitting, and registration requirements at local, state, or federal levels.
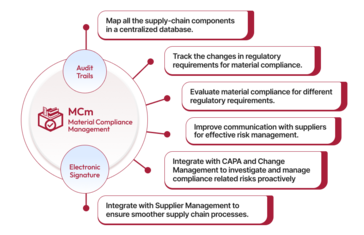
Qualityze Material Compliance Management solution, which is user-friendly and efficient, companies can respond better to changing regulatory requirements across the globe. Additionally, the solution helps stakeholders to view compliance across all supply chain tiers in real-time and understand how risk impacts their goals.
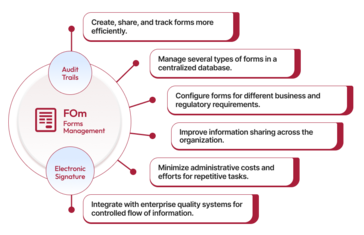
Qualityze Forms Management integrates with your enterprise-quality systems to reduce process cycle time and facilitate the sharing of data where it is needed, simultaneously enhancing the quality and accuracy of the collected data. As a result, productivity will be significantly improved.

As a next-generation Enterprise Quality Management System with robust integration capability, Qualityze ensures that post market surveillance activities including Field Safety Action, Recalls, Complaints Handling and Adverse Events are managed efficiently. With our solution, you can standardize data fields to consistently capture all the required details regarding Field Safety Actions and Recalls. As a result, it will improve decision-making processes. The Qualityze Field Safety & Recall Management solution enables enterprises to continuously analyze and improve their field safety & Recall management processes to keep consumers safe.
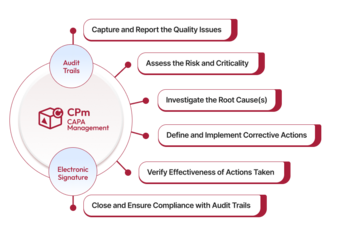
Qualityze CAPA Management enables organizations to develop a risk-based, compliance-driven process for addressing systemic issues to prevent recurrence. It guides the user through a comprehensive approach for mitigating quality issues while meeting compliance and fostering a culture of continuous improvement.
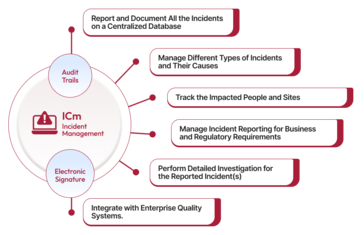
Qualityze Incident Management empowers you to proactively identify and handle near-miss events, manage incidents effectively, and efficiently report on compliance. This solution streamlines all aspects of incident management, including documentation, identification, organization, prioritization, investigation, escalation, resolution, and review. By establishing proper controls, you can prevent many workplace incidents, reduce your company’s liabilities, resolve incidents in a timely manner, and maintain compliance with standards like ISO and OHSAS guidelines. Most importantly, you will earn the trust of your employees. Brand building goes beyond creating a positive customer experience; it also involves fostering a positive and safe work culture.
Qualityze Reviews

Ankur V.
Assistant VP at Credit SuisseGreat for Archiving and Document Tracking
John M.
Qualityze for the Medical Device Industry
Moira B.
Dynarex Testimonial for Qualityze
About
What is Qualityze?
Qualityze is an AI-powered, cloud-based Quality Management System (QMS) built natively on Salesforce for Life Sciences and Manufacturing organizations that need to streamline quality processes and maintain continuous compliance. The platform provides enterprise-grade security, scalability, and real-time visibility across the entire quality lifecycle. With Qualityze QMS, quality, regulatory, and safety teams can automate critical workflows, reduce manual effort, and accelerate decision-making while staying audit-ready. The unified, modular platform connects key quality processes in one system, eliminating data silos and improving traceability from issue identification through resolution. The solution supports essential quality processes including CAPA, nonconformance management, document control, training management, audit management, change management, supplier quality, inspection management, complaints handling, field safety, calibration, maintenance, incident management, permit management, forms management, and structured 8D problem solving, along with centralized master data management and secure third-party integrations through Qualityze Connect. Purpose-built for highly regulated environments, Qualityze supports global compliance requirements such as FDA, ISO 13485, and GxP. Its flexible and configurable architecture enables organizations to scale quality operations efficiently without heavy customization. Organizations choose Qualityze for its Salesforce-native foundation, AI-powered automation, unified end-to-end QMS platform, and rapid deployment capabilities—helping teams achieve a measurable quality and compliance advantage.